Trending...
- Tacoma Dome Welcomes Class of 2026
- From Broken to Soaring Week 40
- CCHR Calls Out Psychiatry's Pattern of Resistance to Antidepressant Deprescribing
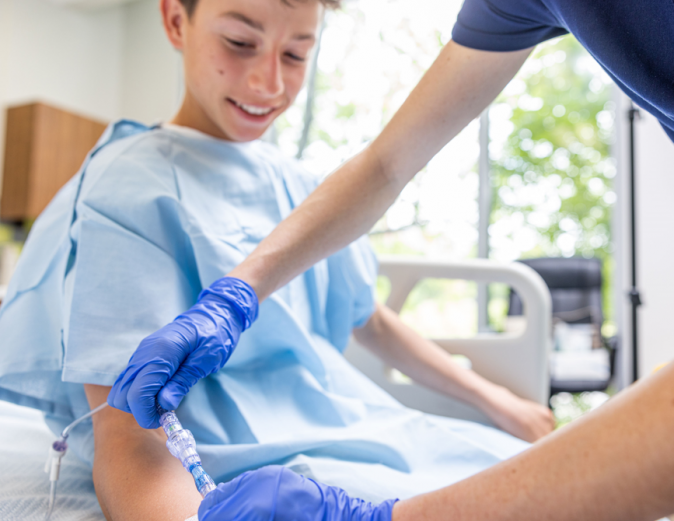
FAYETTEVILLE, Ark. - Washingtoner -- Lineus Medical, a medical device company focused on reducing IV complications, announced that the company's flagship product, SafeBreak Vascular, has received clearance from the U.S. Food and Drug Administration (FDA) for use in pediatric patients. SafeBreak Vascular has been shown in adult clinical studies to reduce IV complications with its innovative breakaway technology1.
SafeBreak Vascular is the first breakaway device cleared by the FDA and proven to reduce IV complications in adults by 44%1. It fits between the IV tubing from the IV pump and the patient's IV access point at the arm. When there is a damaging force on the line, SafeBreak separates to protect the patient's IV. This innovative feature helps prevent accidental dislodgement of the catheter, as well as other IV complications caused by damaging forces on the IV line1. This is especially important for pediatric patients who may be more active, less inclined to follow instructions, and less aware of their surroundings.
More on Washingtoner
"We are incredibly proud to receive clearance from the FDA for the use of SafeBreak Vascular with pediatric patients," said Vance Clement, CEO of Lineus Medical. "SafeBreak provides a simple, effective solution for preventing IV complications, which reduces IV restarts and additional needlesticks for patients. Now, it doesn't matter if the patient is 1 or 100 years old, SafeBreak is cleared for use on all peripheral IVs. Our goal is to have a SafeBreak on every IV line, everywhere in the world, and this additional clearance lets us help one of the most vulnerable populations that needs it the most, our kids."
ABOUT LINEUS MEDICAL
Lineus Medical is the developer of a unique, patented, break-away technology that is designed to work with all types of medical tubing. Complications with feeding tubes, IV lines, urinary catheters and any other type of medical tubing may be reduced as breakaway devices separate to protect the patient. More information about Lineus Medical can be found on the company website: www.lineusmed.com or you can follow the company on Twitter or LinkedIn.
1 Dipper Study Data on File
MKG-0194 REV 00
SafeBreak Vascular is the first breakaway device cleared by the FDA and proven to reduce IV complications in adults by 44%1. It fits between the IV tubing from the IV pump and the patient's IV access point at the arm. When there is a damaging force on the line, SafeBreak separates to protect the patient's IV. This innovative feature helps prevent accidental dislodgement of the catheter, as well as other IV complications caused by damaging forces on the IV line1. This is especially important for pediatric patients who may be more active, less inclined to follow instructions, and less aware of their surroundings.
More on Washingtoner
- What Would you Do with Your Time if it Was Actually Money?
- Mr. Hospital Bed Showcases the Best Hospital Bed and Air Mattress for Bed Sores for 2026
- City of Tacoma Presents Updated Financial Forecast as Next Step in 'Roadmap to Recovery' to Navigate National Economic Pressures
- Tacoma: Full Intersection Closure at E. 11th Street and St. Paul Avenue for One-Day Asphalt Repairs on June 27
- Spokane: Early-Morning House Fire Damages Two Homes on East Sanson Avenue
"We are incredibly proud to receive clearance from the FDA for the use of SafeBreak Vascular with pediatric patients," said Vance Clement, CEO of Lineus Medical. "SafeBreak provides a simple, effective solution for preventing IV complications, which reduces IV restarts and additional needlesticks for patients. Now, it doesn't matter if the patient is 1 or 100 years old, SafeBreak is cleared for use on all peripheral IVs. Our goal is to have a SafeBreak on every IV line, everywhere in the world, and this additional clearance lets us help one of the most vulnerable populations that needs it the most, our kids."
ABOUT LINEUS MEDICAL
Lineus Medical is the developer of a unique, patented, break-away technology that is designed to work with all types of medical tubing. Complications with feeding tubes, IV lines, urinary catheters and any other type of medical tubing may be reduced as breakaway devices separate to protect the patient. More information about Lineus Medical can be found on the company website: www.lineusmed.com or you can follow the company on Twitter or LinkedIn.
1 Dipper Study Data on File
MKG-0194 REV 00
Source: Lineus Medical
0 Comments
Latest on Washingtoner
- Mike Williams Golf Center Now Open at Georgia's Lanier Islands Resort
- Appliance EMT Launches June "Summer Rescue" Promotion
- New Luxury Single Family Homes From $976,990 in Manalapan
- Longevityresearch.ca Unveils a Unique Bayesian Causal Atlas; Saves up to 7.9 life years/patient
- K2 Integrity Acquires RiskFront AI to Deliver AI Automation for Financial Crime Compliance and Risk Operations
- HousingWire acquires Keeping Current Matters, putting local market data into the tools agents use to win listings
- KIDZONET & Ocean Telecom Launch UK First eSIM Child Protection — EasySim AI Safe SIM Cards
- School Dental Screening Programs Conducted in Dubai
- British Brand Daniel Mason™ Expands Premium Braided Leather Belt Collection Internationally
- Looking for expert pool tiling in Gold Coast? Call Avid Tiling
- SPD Investigate Homicide In North Spokane - One Male In Custody
- Hosted Network Powers National Growth with netElastic vBNG, CGNAT and netVision
- Tacoma: Hylebos Bridge to Close for Five Hours on June 11 for Centerlock Replacement
- Super Lawyers Recognizes Inman & Tourgee Attorneys Mark Tourgee and Jacob Rinn
- PropAccount.com Launches PropGenie, the First Branding Studio Built for Prop Firm Operators
- Rushing Headlong: Health IT's Legacy and the Road to Responsible AI is named 2025 Foreword INDIES Book of the Year Awards Winner
- Washington State High School Students Launch Virtual Rocketry Summit
- The Problem With AI Isn't Compute. It's Memory
- Golden Visa Countries Outpace Eurozone Growth Over Eight Years, New La Vida Analysis Finds
- Allstream Energy Partners Announced as Official Media Partner for the 2nd Annual Permian Power Conference